when BrO3^ ion react with Br^ ion in acidic medium ,Br2 is liberated.the equivalent mass of Br2 in the reaction is (M=molar mass of Br2) (1)5/3M (2)3/5M (3)4/6M (4)5/8M

Question Video: Writing a Net Ionic Equation for the Reaction of Chlorine Water with Aqueous Sodium Bromide | Nagwa
What will be the concentration of bromide ions on mixing 300 ml of 0.1 M AgNO3 with 200ml of 0.1 M CaBr? - Quora
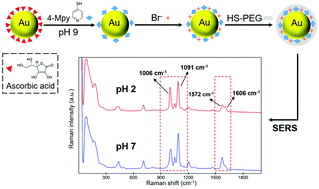
Bromide ion-functionalized nanoprobes for sensitive and reliable pH measurement by surface-enhanced Raman spectroscopy - Analyst (RSC Publishing)
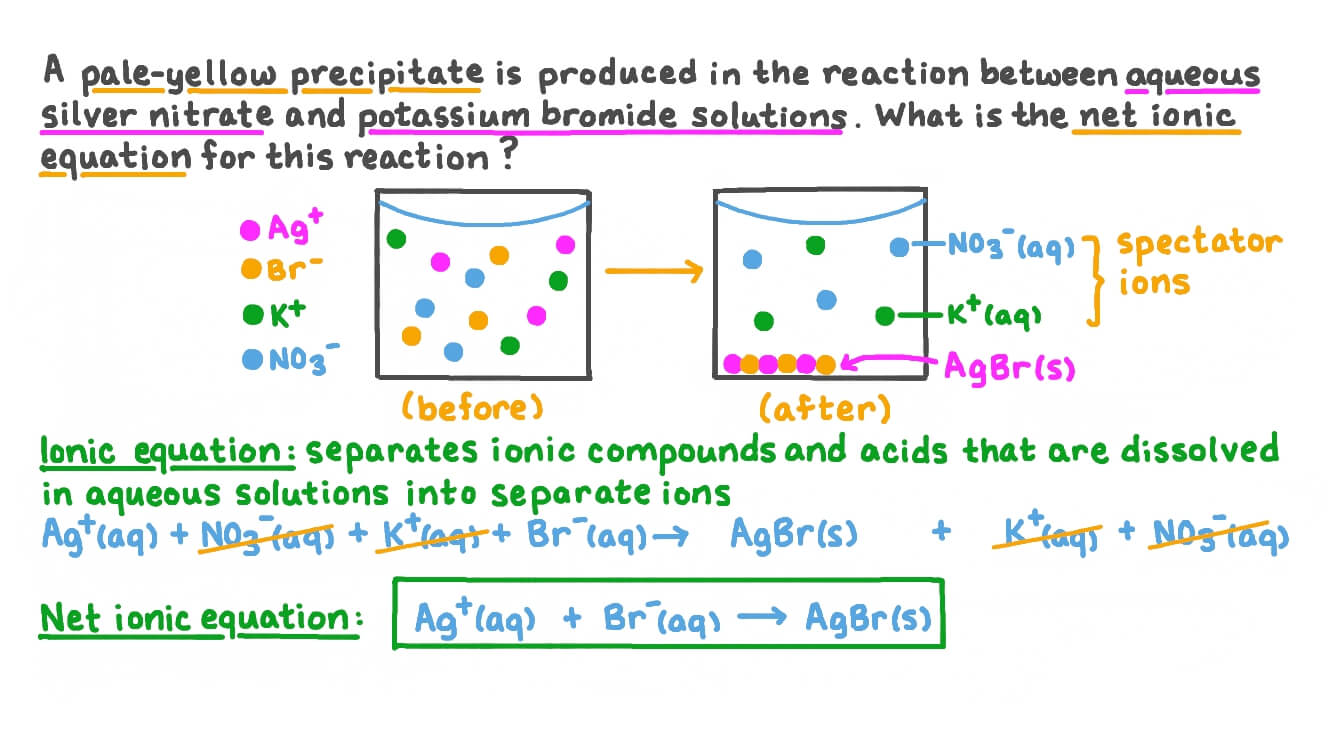
Question Video: Writing a Net Ionic Equation for the Reaction between Silver Nitrate and Potassium Bromide Solutions | Nagwa

The Multiple Role of Bromide Ion in PPCPs Degradation under UV/Chlorine Treatment | Environmental Science & Technology








.jpg)